Immunohistochemistry (IHC) involves the labeling of cellular proteins in tissue samples with a specific primary antibody and visualization of the target with detection reagents. Cell Signaling Technology (CST) offers over 800 antibodies validated for IHC, as well as IHC diluents, detection reagents, substrates and controls, to ensure that your IHC studies yield accurate and reproducible results. The determination of target specificity in immunohistochemical analysis requires multiple validation steps. CST scientists use a variety of methods, as appropriate, to validate each IHC-recommended antibody, ensuring that the staining you observe with each CST antibody is specific and believable.
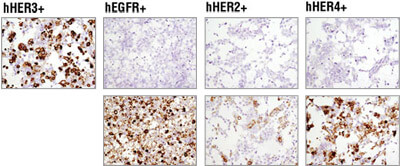
Cell Pellets: Immunohistochemical analysis of paraffin-embedded 293T cell pellets transfected with human erbB family members HER3, EGFR, HER2 and HER4 (from left to right as indicated) using HER3/ErbB3 (D22C5) XP®Rabbit mAb #12708 (top panels). Transfections were confirmed using EGF Receptor (D38B1) XP® Rabbit mAb #4267 (lower left), HER2/ErbB2 (D8F12) XP® Rabbit mAb #4290 (lower middle) and HER4/ErbB4 (111B2) Rabbit mAb #4795 (lower right).
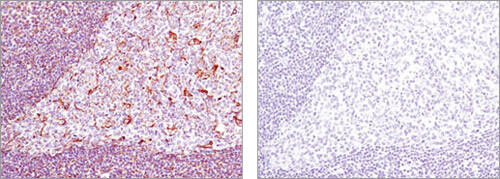
Blocking Peptide: Immunohistochemical analysis of paraffin-embedded human tonsil using Vimentin (D21H3) XP® Rabbit mAb #5741 in the presence of control peptide (left) or antigen-specific peptide (right).
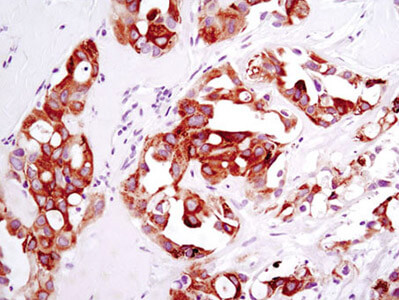
Xenograft: Immunohistochemical analysis of paraffin-embedded HCC78 xenograft using ROS1 (D4D6) Rabbit mAb #3287.