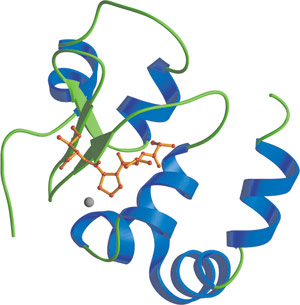
The third BIR domain of XIAP, showing zinc atom and binding of Smac N-terminal residues (red).
The Baculovirus IAP Repeat (BIR) domain is an approximately 70 amino acid zinc-binding domain that was first identified through sequence homology among proteins belonging to the Inhibitors of Apoptosis (IAP) family. Present in one to three tandem copies per protein, the BIR domain has been identified in over 80 different proteins in eukaryotic organisms. Most of what is known about BIR domains comes from their role in IAP proteins. IAPs bind to and inhibit caspases, a class of cysteine proteases involved in propagating apoptotic signals within the cell. The BIR domain is necessary for IAP protein interaction with a number of proapoptotic factors, including invertebrate death inducers (i.e. Reaper, Grim, HID and Doom of Drosophila) and vertebrate and invertebrate caspase proteases. In inhibiting caspase-9, the third BIR domain of XIAP interacts with an amino-terminal ATPF/AVPY motif found on the linker peptide on the p12 small subunit of caspase-9, which becomes exposed after procaspase-9 proteolytic cleavage. This interaction is regulated by the Smac/Diablo protein, which presents a high affinity BIR3 interacting peptide to prevent for XIAP BIR3 binding and thereby sequester XIAP from caspase-9. The second BIR domain of XIAP exerts an anti-apoptotic effect by acting as a regulatory element for caspase binding, while the amino-terminal linker interacts with and blocks the substrate groove of caspase-3 and -7. This linker peptide lies across the caspase active site in an orientation that opposes substrate binding. Deletion of this second BIR domain abrogates the anti-apoptotic function of XIAP, as removal of this BIR domain prevents the protein from adopting a conformation that would affect caspase binding. Finally, the BIR domain also appears to be capable of mediating homophilic interactions.
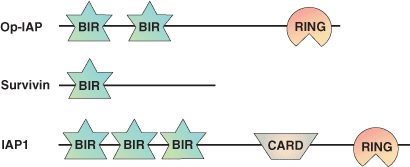
| BIR Domain Proteins | Binding partners |
| Survivin | Survivin via a homotypic interaction |
| Op | HID, Grim, Reaper |
| D-AIP1 | HID, Grim |
| D-AIP2 | Reaper |